CAR-T therapy is an advanced immunotherapy in which a patient’s own T cells are collected, genetically modified to recognize a cancer-related target and then expanded before being reinfused to attack malignant cells.
It has become one of the most important breakthroughs in modern oncology because it turns immune cells into a living therapeutic product. That idea has already shown strong clinical impact in several hematologic cancers and continues to expand into new indications and manufacturing models.
CAR-T therapy reprograms the patient’s own immune cells so they can detect and destroy specific cancer targets more effectively.
What is CAR-T therapy?
CAR-T therapy stands for chimeric antigen receptor T-cell therapy. It is a form of adoptive cell therapy in which T cells are engineered in the lab to express an artificial receptor that helps them recognize and attack cancer cells.
Unlike standard drugs, CAR-T is a personalized cell product. Each batch is manufactured for one patient, which makes the therapy highly specific but also technically demanding.
CAR-T therapy is not a conventional drug, it is a living and highly personalized therapeutic product.
How CAR-T therapy works
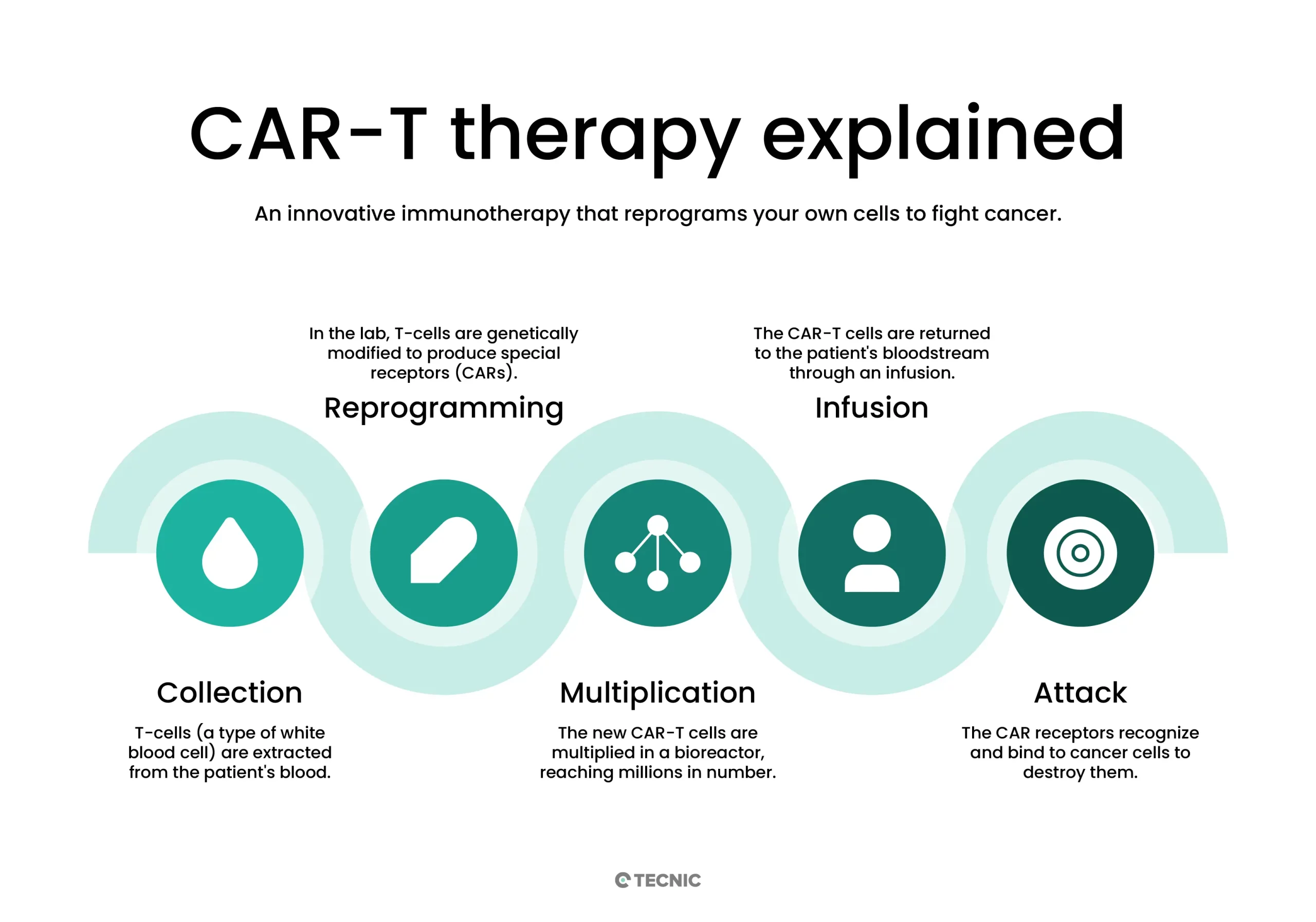
The process usually follows a defined sequence from cell collection to reinfusion. Although the exact workflow can vary, the manufacturing logic is broadly consistent.
T cells are obtained from the patient through leukapheresis.
The cells are engineered to express a CAR that targets a specific antigen on cancer cells.
The modified cells are expanded in controlled culture until they reach the therapeutic dose.
The final CAR-T product is infused back into the patient so the cells can seek and attack the target.
CAR-T therapy is not only an immunotherapy concept, it is also a complex GMP cell-manufacturing workflow.
Five important breakthrough uses of CAR-T therapy
CAR-T therapies have shown their strongest approved impact in hematologic cancers, while newer applications continue to emerge in clinical development.

Why CAR-T therapy is so relevant in modern oncology
CAR-T therapy matters because it has shown that the immune system can be reprogrammed into a highly targeted cancer treatment. That has changed expectations in oncology, especially for patients with relapsed or refractory blood cancers.
High specificity
The engineered receptor helps T cells identify tumor-associated antigens with a much more directed mechanism than conventional systemic therapies.
Living therapy logic
CAR-T is not just administered, it can expand, persist and continue functioning as an active cellular product after infusion.
CAR-T therapy is clinically powerful, but its full impact depends as much on manufacturing and logistics as on immunology itself.
Manufacturing and scaling challenges in CAR-T therapy
CAR-T manufacturing remains one of the biggest barriers to broader access. Each batch is personalized, the starting material varies from patient to patient, and the process requires tightly controlled environments, robust quality systems and reliable expansion platforms.
Each product is made for one patient, which limits the simplicity of large-scale standardization.
The starting T-cell population can differ significantly depending on disease and prior treatment history.
Isolation, activation, modification, expansion and release testing all require tightly controlled conditions.
Closed systems, advanced bioreactors and stronger digital traceability are becoming essential to scale the field more effectively.

How TECNIC fits this workflow
TECNIC fits this topic directly because CAR-T therapy depends on controlled cell expansion, reproducible manufacturing conditions and scalable process design. As CAR-T moves toward broader clinical availability, expansion platforms and process control become increasingly important.
Bioreactors
Relevant when CAR-T and other advanced cell therapies need controlled expansion from development to GMP manufacturing.
Cell and gene therapy context
CAR-T therapy fits naturally within the broader advanced-therapy field already reflected in TECNIC content.
Software and control
User control, batch traceability and repeatable execution are increasingly important in CAR-T manufacturing.
Contact TECNIC
When advanced cell therapy workflows need stronger expansion control and scale-up logic, direct technical discussion becomes more useful than theory alone.
This article works best when CAR-T therapy is framed as both a major clinical breakthrough and a demanding manufacturing process.
Frequently asked questions
What is CAR-T therapy?
It is an advanced cell therapy in which a patient’s T cells are genetically modified to recognize and attack cancer cells.
How does CAR-T therapy work?
T cells are collected, engineered to express a chimeric antigen receptor, expanded in the lab and reinfused into the patient.
What cancers is CAR-T therapy used for?
It is mainly used in certain hematologic cancers such as acute lymphoblastic leukemia, diffuse large B-cell lymphoma, mantle cell lymphoma and multiple myeloma.
Is CAR-T therapy used in solid tumors?
It is being actively studied, but solid tumors remain more challenging and no equivalent broad success has yet matched the main blood-cancer indications.
What is the biggest challenge in CAR-T therapy?
One of the biggest challenges is manufacturing, especially producing personalized cell products quickly, consistently and at broader scale.
Exploring how CAR-T therapy connects with scalable cell expansion and process control?
Explore TECNIC’s bioprocess solutions or speak with our team to review the right setup for advanced cell-therapy manufacturing workflows.